
This product line focuses on providing Class II medical device-level hardware manufacturing solutions with Internet of Things (IoT) connectivity for the fields of remote monitoring, diagnostic support and personalized treatment. Its core is to solve the complex challenges of medical-grade data acquisition accuracy, long-term biocompatibility, and complete design control and production traceability in compliance with regulations such as FDA 510(k) or CE MDR. As a vertically integrated manufacturer, we will serve global high-end customers (such as as a supplier of heraeus medical components manufacturer) The stringent standards accumulated throughout the entire process from medical device injection mold development to sterile packaging.
JinYi Mold not only provides equipment manufacturing, but also engineering services based on ISO 13485 medical device quality management system. From the product definition stage, we collaborate with customers on risk analysis (such as ISO 14971), usability engineering (UE) and electromagnetic compatibility (EMC) design to ensure that the product's hardware foundation meets the fundamental requirements for reliability and safety of medical devices while achieving real-time data upload (such as HL7/FHIR protocol), low-power Bluetooth (BLE) connection and App control.

Key points of engineering technology
Mold engineering:
The device shell and contact parts are strictly made of USP Class VI or ISO 10993 biocompatibility certified materials, such as medical-grade PC, ABS, and PPSU. Our medical component plastic mold uses highly polished corrosion-resistant steel (such as S136H), and uses a vacuum injection molding process to ensure that complex sensor cavities, micro-channels and other structures are filled without bubbles, ensuring functional integrity and meeting cleaning verification requirements.
Electronic system integration:
The equipment integrates medical-grade high-precision sensors (such as pressure, photoelectricity, temperature), and the signal chain is shielded and filtered. Our appliance medic mold manufacture capabilities ensure sensors are precisely positioned and packaged. As a system integrator of medical device components, we provide a complete set of electronic solutions from medical PCBA design (meeting IEC 60601-1 safety regulations), firmware development to data encryption and privacy protection (meeting GDPR/HIPAA requirements).
Verification system:
The design history file (DHF) and device master record (DMR) we established ensure full traceability. Products must pass a complete verification process including accuracy and repeatability verification (compared to gold standard equipment), environmental reliability testing (such as high and low temperature, humidity and heat), mechanical durability testing (such as drop, key life), and strict EMC radiation and immunity testing.

Validation and Quality Assurance
Our quality system is built around medical device regulations to ensure successful product registration.
Design and process validation:
Perform complete design verification (DV) and process qualification (PQ). The mold is optimized through mold flow analysis, and multiple rounds of trial mold samples from T0 to T2 are produced for functional, assembly and accelerated aging testing before freezing.
Product performance and testing:
We can assist in sending the finished product to a third-party laboratory with ISO 17025 qualifications for full performance testing, biocompatibility testing (cytotoxicity, sensitization, irritation) and packaging integrity verification.
Regulatory document support:
We can provide the technical documents required to support FDA QSR 820 or CE MDR submissions, including material certification, process validation reports, cleaning validation reports and complete risk management documents.

Delivery and support
JinYi Mold provides ODM/OEM turnkey services from active medical device product development, mold manufacturing, injection molding, clean workshop assembly (such as Class 10,000 or Class 100,000) to final sterilization (such as EO ethylene oxide) and packaging. The production line is equipped with an MES manufacturing execution system to realize full-link traceability of single products from raw materials to finished products.
If you are developing the next generation of IoT Medical Devices and need a partner with deep experience and track record of success in medical device compliance, precision mold manufacturing, electronic system integration and rigorous quality management systems, we can provide comprehensive solutions from concept to market access. Please provide your product requirements and target markets, and we will conduct a preliminary regulatory and manufacturing feasibility assessment.
About Us

Jinyi Mold was established in 1998 and is headquartered in Dongguan, Guangdong. Our professional team provides injection mold and die-casting mold services, providing you support in every aspect from design, manufacturing to final assembly.
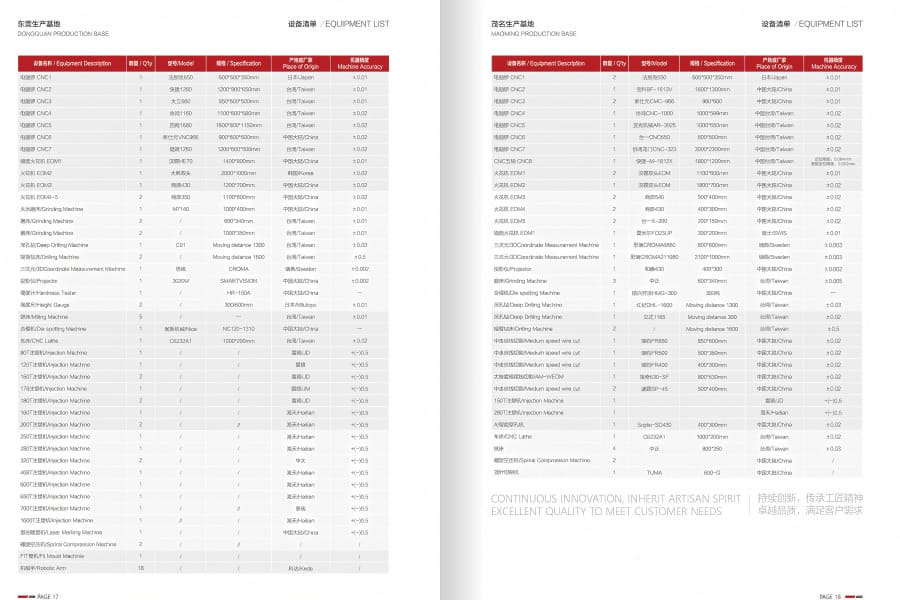
Production equipment:
dozens of CNC machining centers (gantry & 5-axis), injection molding machines (80-1600 tons), EDM machines, automation equipment, etc.
Scope of services:
Establish solid partnerships with Europe, Australia and Asia to carry out international export business.
Company production capacity: A 13,000㎡ production base in Maoming (built in 2022), backing an annual turnover surpassing 150 million RMB.
Honors and qualifications:
Possess High-Tech Enterprise Certification, ISO9001:2015 Quality management system certification.

Logistics and freight:
Logistics follows international transportation standards, and packaging is packed in reinforced wooden boxes, filled with PE foam and wrapped in moisture-proof bags.

Marketing: Selina Chan
WhatsApp:+8618969686504
Email:selina@jy-mould.com
Contact us to discuss how we can support your project needs
Please know that when you choose Jinyi Mold, we are not only a supplier, but also a Chinese manufacturing partner with 25 years of experience in mold design, manufacturing, and assembly.













