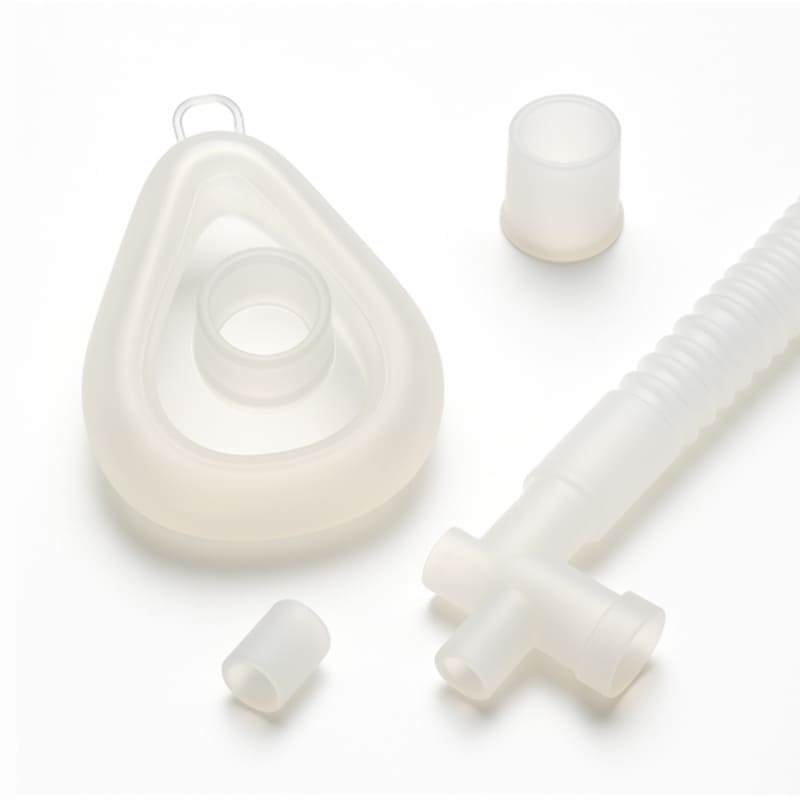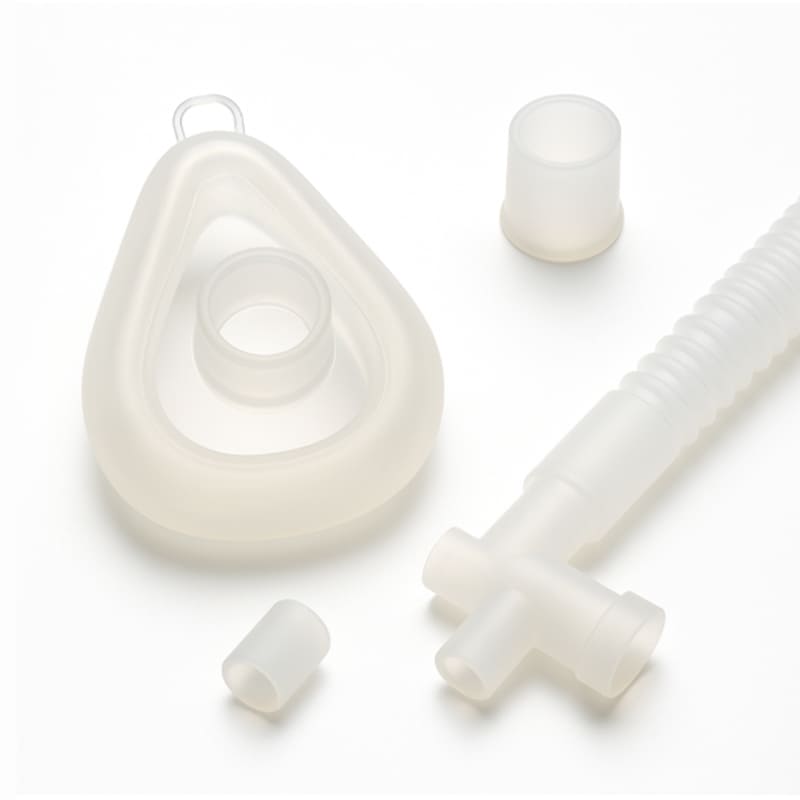
JinYi Mold focuses on medical-grade silicone casting and overmolding, providing key silicone components for diagnostic equipment, respiratory care, wearable health monitoring and surgical instruments and other fields. The key to medical applications is to go beyond conventional precision to ensure full biocompatibility, long-term material stability and strict regulatory compliance. We focus on overcoming core engineering challenges such as firmly bonding flexible silicone to rigid plastic/metal substrates, achieving microfluidic channel sealing, and maintaining component performance over multiple sterilization cycles.
Relying on its rich experience in the field of precision mold manufacturing, JinYi Mold integrates the ideas of ISO 13485 medical device quality management system into every aspect of the production of medical silicone parts. Our materials scientists work closely with our engineering teams to provide global medical device manufacturers with integrated, compliant and reliable solutions from rapid prototyping to mass production, from material selection, mold development, clean room manufacturing to sterilization validation support.

Key points of engineering technology
Materials and process systems oriented toward medical safety:
We focus on the use of platinum-cured liquid silicone (LSR), which has extremely low extractables. This characteristic complies with stringent standards such as ISO 10993 biocompatibility and USP Class VI. For silicone overmolding of medical device shells, we are proficient in surface pretreatment and bonding technology to ensure a stable and sealed interface between silicone and base materials such as ABS, PC, stainless steel, etc., without the risk of delamination.
High-precision medical molds and micro-molding technology:
Molds used in silicone casting of medical equipment have extremely high requirements for cavity accuracy and surface finish. We use mirror steel and a full hot runner system, and use vacuum-assisted technology to manufacture extremely precise medical biocompatible silicone parts, such as microvalves, thin-walled sealing films, catheter interfaces, etc., and can effectively prevent bubble defects.
Sterility guarantee and value-added secondary processing:
We provide comprehensive post-processing services, including: surface coating to achieve antibacterial or marking functions, laser marking for product traceability, and sandblasting to improve tactile and optical performance. We understand the needs of the sterile supply chain and can provide proven cleaning, packaging and sterilization (such as EO, gamma ray) process support.

Quality Assurance and Verification
We implement the most stringent verification procedures in the medical device industry to ensure that each product is traceable and has stable performance.
Process verification and design control:
Starting from DFMEA (Design Failure Mode Analysis), use DOE (Design of Experiments) to optimize and determine all key process parameters (mixing ratio, injection speed, curing temperature and duration), build a stable process window and complete verification (IQ/OQ/PQ).
Compliance and comprehensive performance testing:
Not only do products undergo full inspection of size (using optical measurement) and appearance, we can also assist or provide cytotoxicity testing, skin sensitization testing, physical and chemical performance testing, and aging testing to simulate service life, etc. to assist customers in regulatory registration.
Document management and strict batch control:
We implement a complete batch record (DHR) system to ensure that the entire process from raw materials to finished products can be traced, and can provide a complete technical document package that meets the requirements of the quality system.

Support and delivery
We understand the importance the healthcare industry places on supply chain reliability. Depending on the end use of the product, you can choose to use primary packaging (such as dialysis paper, blister boxes), vacuum bags or clean bags for packaging, and provide complete sterilization certificates, material compliance statements and test reports. As a Chinese supplier focusing on precision manufacturing, we rely on our rapid response capabilities, open project management and stable production capabilities to help customers meet their needs at all stages from clinical trials to global commercial production.
JinYi Mold strives to become an indispensable R&D and manufacturing partner for high-end silicone components for medical device manufacturers, transforming complex medical designs into safe, efficient and mass-produced products.
About Us

Jinyi Mold was established in 1998 and is headquartered in Dongguan, Guangdong. Our professional team provides injection mold and die-casting mold services, providing you support in every aspect from design, manufacturing to final assembly.
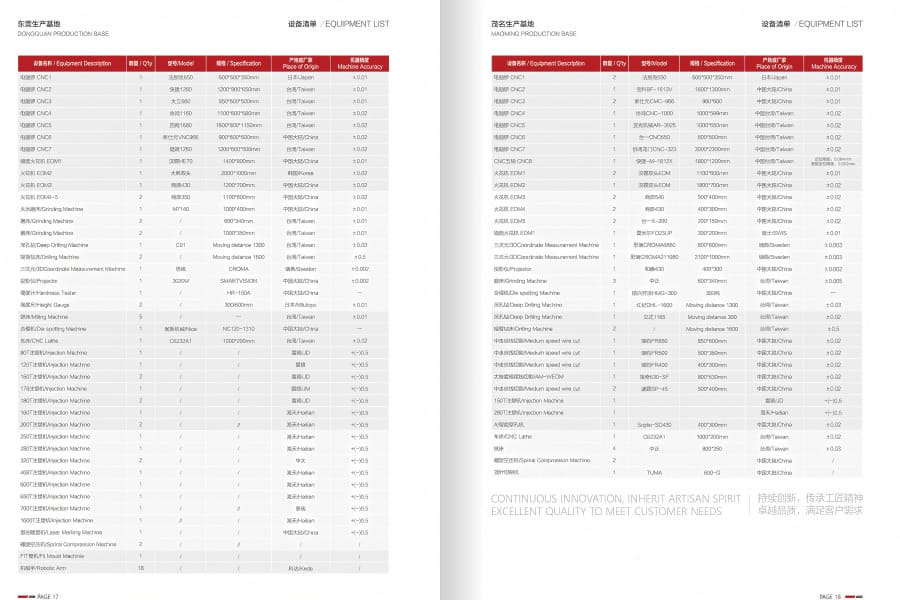
Production equipment:
dozens of CNC machining centers (gantry & 5-axis), injection molding machines (80-1600 tons), EDM machines, automation equipment, etc.
Scope of services:
Establish solid partnerships with Europe, Australia and Asia to carry out international export business.
Company production capacity: A 13,000㎡ production base in Maoming (built in 2022), backing an annual turnover surpassing 150 million RMB.
Honors and qualifications:
Possess High-Tech Enterprise Certification, ISO9001:2015 Quality management system certification.

Logistics and freight:
Logistics follows international transportation standards, and packaging is packed in reinforced wooden boxes, filled with PE foam and wrapped in moisture-proof bags.

Marketing: Selina Chan
WhatsApp:+8618969686504
Email:selina@jy-mould.com
Contact us to discuss how we can support your project needs
Please know that when you choose Jinyi Mold, we are not only a supplier, but also a Chinese manufacturing partner with 25 years of experience in mold design, manufacturing, and assembly.