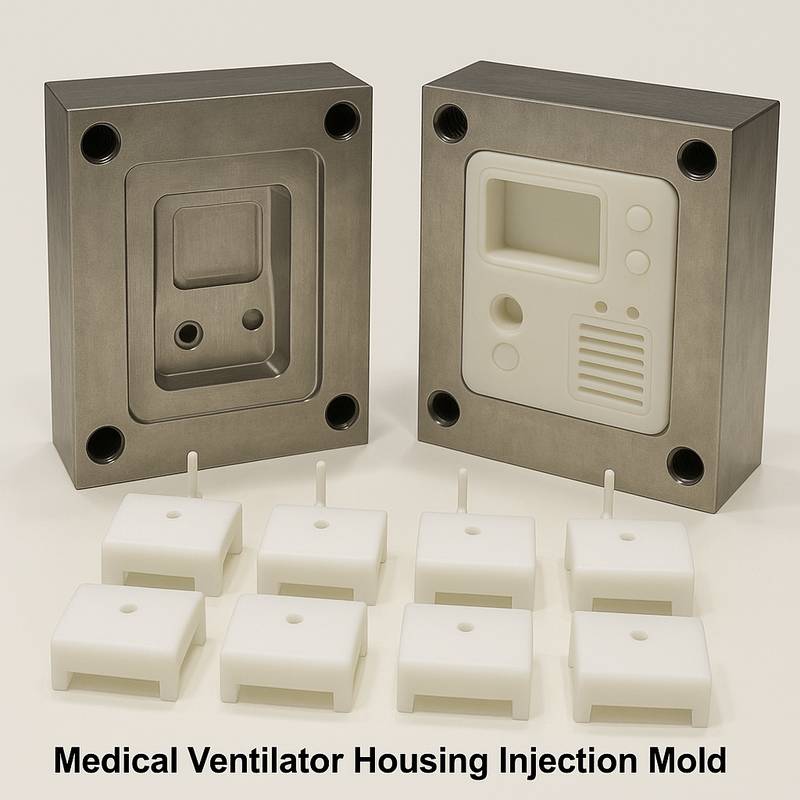
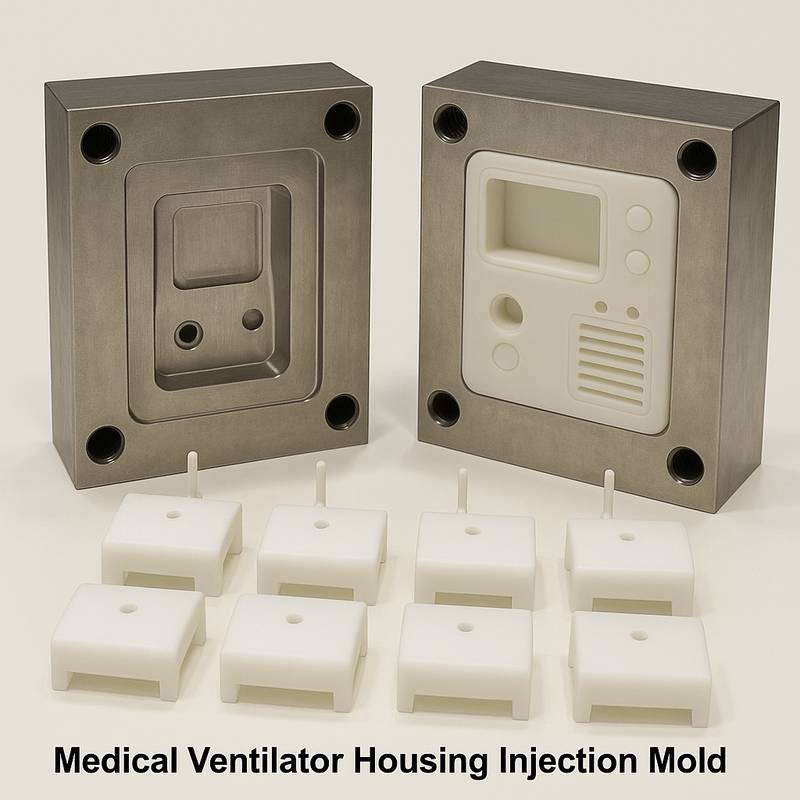
This series of high-precision molds from JinYi Mold is specially developed and designed for the shell and structural components of medical ventilators and other critical life support equipment. Ventilator equipment places extremely stringent requirements on the airtightness, structural strength, long-term stability and biocompatibility of plastic components. The core of the mold is to ensure that each shell produced can achieve zero-leakage sealing, precise component assembly, and durability against repeated sterilization during a use cycle of tens of thousands of hours. These are directly related to the safety and reliability of medical equipment.
As a professional medical shell injection mold manufacturer, JinYi Mold has integrated the ISO 13485 quality management system serving global medical brands into the entire mold development process. We not only provide molds but also provide solutions covering material certification, clean production and verification support, and are committed to transforming the design intentions of medical device manufacturers into stable, compliant products that can be mass-produced.
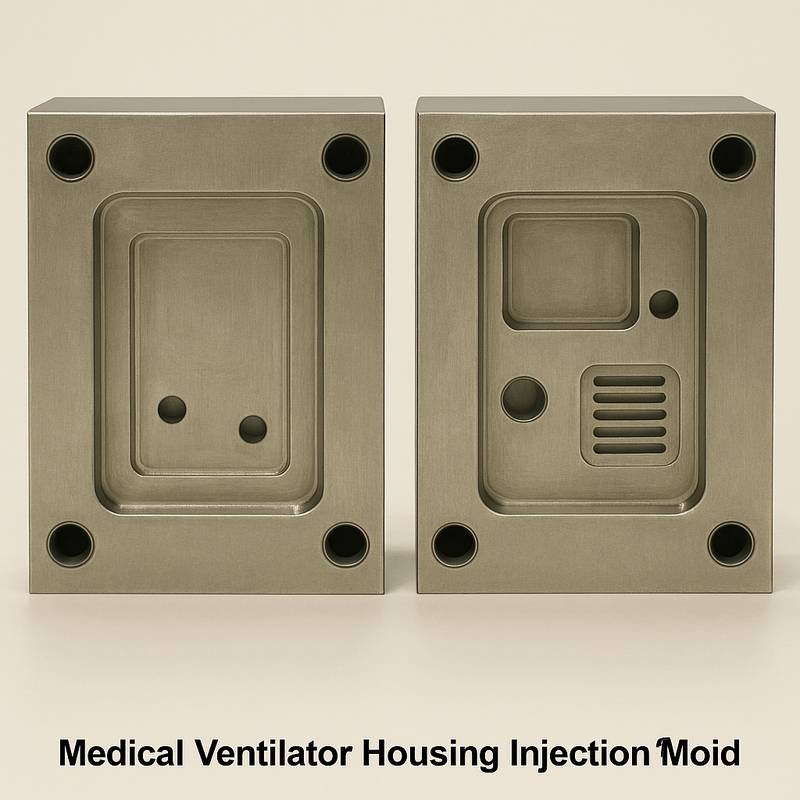
Key points of systematic engineering technology
Design and material collaboration for medical safety:
From the beginning of the project, our engineering team conducted manufacturability analysis based on the customer's design to ensure uniform wall thickness and eliminate stress concentration points. We work with the world's top material suppliers such as SABIC and Covestro to develop medical-grade PC, ABS and PC/ABS alloys that meet ISO 10993 biocompatibility standards to ensure that the materials have high transparency, impact resistance and chemical sterilization resistance, ensuring the long-term safety of medical ventilator plastic components from the source.
Medical-grade precision mold manufacturing:
S136, NAK80 and other mirror-effect and corrosion-resistant mold steels are used as the core of the mold. With the help of high-precision CNC and EDM processing technology, the tolerance of the key mating surfaces is strictly controlled within ±0.01mm. We specialize in the design of multi-cavity high-speed injection molds. While improving production efficiency, we rely on balanced hot runners and cooling systems to ensure a high degree of consistency in the parts produced in each cavity and meet the stringent requirements for ultra-high stability of plastic injection molds for medical equipment.
Fully automatic closed-loop production process:
In the production process, we use all-electric injection molding machines to achieve millisecond-level precise control of injection speed, holding pressure and cooling time, ensuring that products have consistent surface quality and internal structure. The production line is equipped with real-time temperature control and dehumidification and drying systems, providing an ideal environment for the processing of high-performance engineering plastics and ensuring stable mass production of medical shell plastic injection mold products.
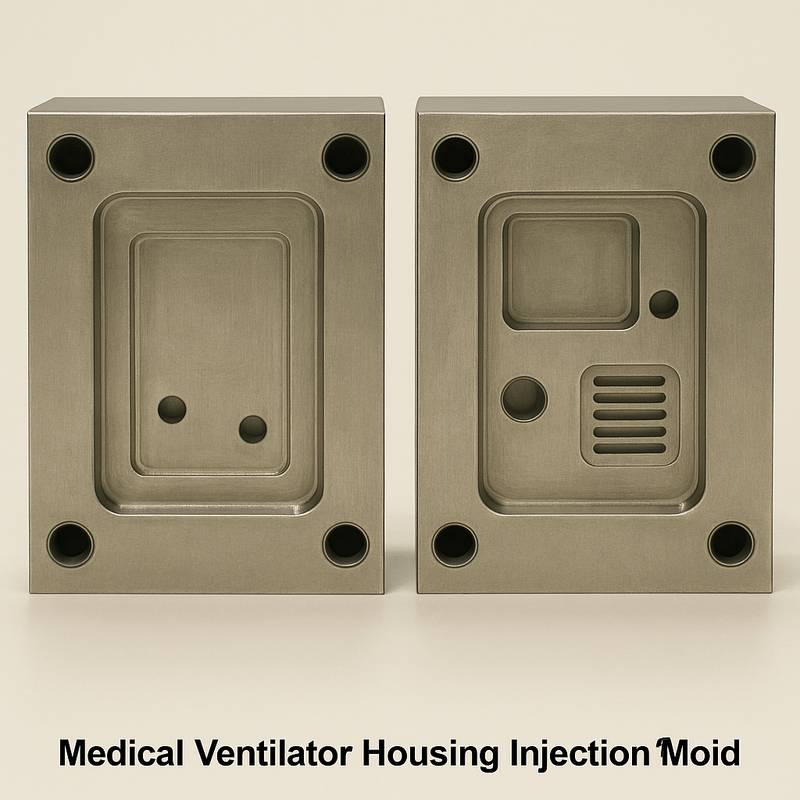
Validation and Quality Assurance
We implement stricter medical product verification standards than general plastic parts and build multiple lines of quality defense.
Full-process parameter monitoring and traceability:
From raw material storage to finished product delivery, all key process parameters are recorded in detail and can be traced, ensuring that the production process is fully controlled and reproducible.
Comprehensive finished product performance testing:
All ventilator shell components are required to pass a series of inspections including three-dimensional dimensional inspection (CMM), air tightness testing, drop testing, and aging resistance evaluation. We can assist in providing third-party authoritative test reports to verify that products comply with international medical standards with objective data.
Sterility and packaging verification support:
We understand the sterilization requirements of medical terminals, can provide verified clean packaging solutions, and provide compatibility support for customers’ subsequent sterilization processes (such as EO sterilization).
Delivery and support
Our molds and production services have successfully supported many well-known medical brands in Europe and the United States, and our products are widely used in ICU equipment, home care and portable medical solutions. Whether it is rapid prototyping for design verification or large-scale commercial production, we can provide flexible, efficient and cost-effective one-stop medical device plastic injection mold solutions.
If you are looking for a reliable medical shell injection mold manufacturer, or plan to develop customized medical ventilator plastic component molds, please contact us. JinYi Mold's professional engineering team will provide you with full support from technical consultation to mass production delivery.
About Us

Jinyi Mold was established in 1998 and is headquartered in Dongguan, Guangdong. Our professional team provides injection mold and die-casting mold services, providing you support in every aspect from design, manufacturing to final assembly.
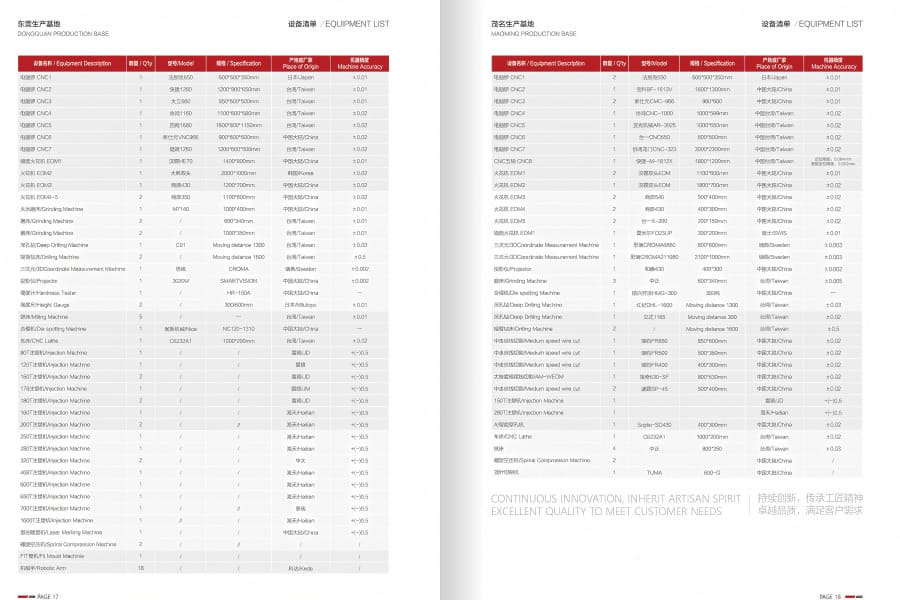
Production equipment:
dozens of CNC machining centers (gantry & 5-axis), injection molding machines (80-1600 tons), EDM machines, automation equipment, etc.
Scope of services:
Establish solid partnerships with Europe, Australia and Asia to carry out international export business.
Company production capacity: A 13,000㎡ production base in Maoming (built in 2022), backing an annual turnover surpassing 150 million RMB.
Honors and qualifications:
Possess High-Tech Enterprise Certification, ISO9001:2015 Quality management system certification.

Logistics and freight:
Logistics follows international transportation standards, and packaging is packed in reinforced wooden boxes, filled with PE foam and wrapped in moisture-proof bags.

Marketing: Selina Chan
WhatsApp:+8618969686504
Email:selina@jy-mould.com
Contact us to discuss how we can support your project needs
Please know that when you choose Jinyi Mold, we are not only a supplier, but also a Chinese manufacturing partner with 25 years of experience in mold design, manufacturing, and assembly.