
This service focuses on rapid prototyping and low-volume manufacturing of highly biocompatible liquid silicone (LSR) components for medical devices, in vitro diagnostic equipment, and wearable medical monitoring products. The core of medical silicone parts is to exceed conventional physical properties and meet regulatory requirements such as ISO 10993 biocompatibility series standards and USP Class VI, ensuring absolute safety and long-term stability when in contact with human tissue or body fluids. We integrate the engineering experience of high and low temperature resistance (-40°C to 200°C), media resistance and long-term sealing accumulated in the silicone casting for automotive gaskets and custom silicone car seals and housings projects, as well as the pursuit of precision appearance and delicate touch in silicone casting for consumer electronics, and apply it to the medical field, providing full-process compliance solutions from material certification, clean room production to sterilization verification support.
Through low cost tooling for short-run production mode, JinYi Mold uses 3D printing master molds and room temperature vulcanization (RTV) silicone molds to deliver functional prototypes for design verification, preclinical research or registration application within 7-14 days, greatly accelerating the product launch process.

Material control
Specializing in the use of platinum-cured liquid silicone, its low extractable properties meet stringent biocompatibility requirements. We work with certified suppliers to provide specialty medical-grade LSR materials ranging from Shore A 10 to more than 80 hardnesses, transparent or opaque, and specific fluorescent imaging. All material batches are provided with complete traceable certification documents.
Drawing on the material development experience of custom silicone car seals and housings, we can tailor the formulation to address specific requirements in medical scenarios such as resistance to gamma/ethylene oxide sterilization, resistance to specific disinfectants, or resistance to long-term immersion in body fluids.
Prototype trial production process
Based on the 3D data provided by the customer, high-precision stereolithography (SLA) 3D printing is used to manufacture the master mold, with a detail resolution of up to 0.05mm. Durable RTV silicone molds are then manufactured and vacuum molded to replicate extremely complex geometries, microfluidic channels or diaphragm structures as thin as 0.3mm.
This low cost tooling for short-run production method is particularly suitable for producing trial batches of 50-500 pieces for functional testing, animal experiments or limited clinical use without having to bear the high cost of steel molds and long delivery times.
Production and processing
Prototyping and trial production are completed in a controlled, clean environment. We offer professional post-processing including: precision trimming to remove the mold line, surface plasma treatment to achieve hydrophilic or hydrophobic properties, and laser marking for product identification and traceability.
We understand the consumer-grade precision assembly requirements that custom silicone phone accessories represent and apply this precision standard to medical components to ensure their precise fit with sensors, plastic housings, or metal needle tubes.

Quality assurance
We follow a rigorous process for medical device product development and provide supporting evidence for prototypes that meet quality system requirements.
Process and mold verification:
Measure and verify the replication accuracy of each set of RTV silicone molds, and record key mixing, vacuum, and curing parameters to ensure process consistency.
Prototype performance characterization:
In addition to dimensional testing, we can assist customers in sending prototypes to laboratories with ISO 17025 qualifications to conduct ISO 10993 series tests such as cytotoxicity, skin sensitization, and irritation, or conduct basic functional (such as flow, sealing) verification.
Sterilization compatibility assessment:
Based on the sterilization method planned by the customer (such as EO, gamma ray, high-pressure steam), a preliminary assessment and suggestions on material compatibility can be provided.
Delivery and support
JinYi Mold provides highly flexible low cost tooling for short-run production services that accurately match the characteristics of medical product development cycles, multiple iterations, and strict regulations. Whether it is silicone grips for innovative surgical instruments, waterproof seals for continuous glucose monitors, or flexible contact parts in rehabilitation aids, we can respond quickly and provide technical support from design consultation to trial production delivery.
If your medical device project is in the proof-of-concept, prototyping or low-volume clinical trial phase and requires silicone parts that combine biosafety, functionality and rapid delivery, please contact us. Please provide your performance requirements and design ideas, and we will initiate a medical compliance manufacturing feasibility analysis.
About Us

Jinyi Mold was established in 1998 and is headquartered in Dongguan, Guangdong. Our professional team provides injection mold and die-casting mold services, providing you support in every aspect from design, manufacturing to final assembly.
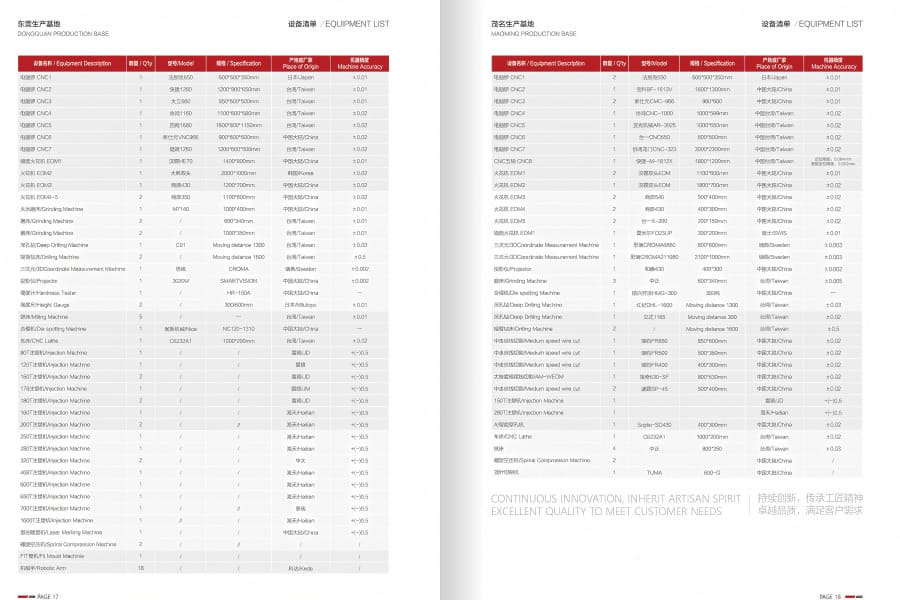
Production equipment:
dozens of CNC machining centers (gantry & 5-axis), injection molding machines (80-1600 tons), EDM machines, automation equipment, etc.
Scope of services:
Establish solid partnerships with Europe, Australia and Asia to carry out international export business.
Company production capacity: A 13,000㎡ production base in Maoming (built in 2022), backing an annual turnover surpassing 150 million RMB.
Honors and qualifications:
Possess High-Tech Enterprise Certification, ISO9001:2015 Quality management system certification.

Logistics and freight:
Logistics follows international transportation standards, and packaging is packed in reinforced wooden boxes, filled with PE foam and wrapped in moisture-proof bags.

Marketing: Selina Chan
WhatsApp:+8618969686504
Email:selina@jy-mould.com
Contact us to discuss how we can support your project needs
Please know that when you choose Jinyi Mold, we are not only a supplier, but also a Chinese manufacturing partner with 25 years of experience in mold design, manufacturing, and assembly.










